
BioPlus Specialty Pharmacy Acquired by Elevance
One of the few 2022 specialty pharmacy acquisitions was announced recently. Elevance Health will acquire long running BioPlus as it seeks to expand its specialty

One of the few 2022 specialty pharmacy acquisitions was announced recently. Elevance Health will acquire long running BioPlus as it seeks to expand its specialty

There is a general understanding that Big Pharma spent a lot of effort and $$$s to counter-detail legislation to allow price negotiations in Medicare. They viewed

We’ve often written about companies that work with hospitals to develop owned-and-operated specialty pharmacies. Those pharmacies boast that they that are significantly better than independent

Walgreens….. its been good to know ya! That’s not saying that Walgreens is going away. To the contrary, it is the dawn of a new day
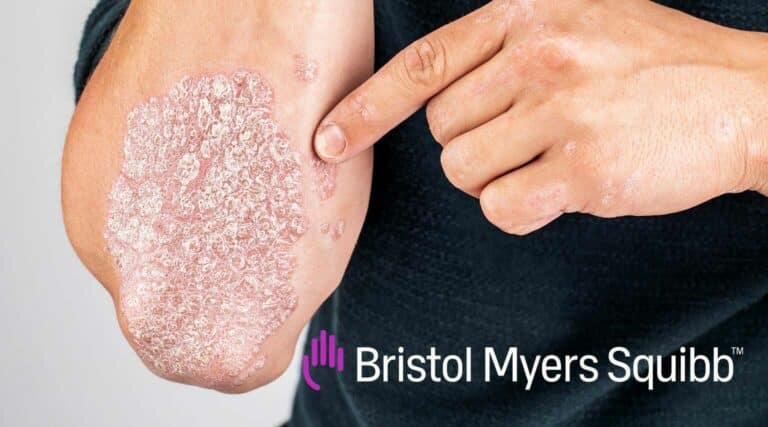
The FDA recently approved a new first-in-class tyrosine kinase 2 (Tyk2) inhibitor, ORAL therapy, Sotyktu (deucravacitinib) from Bristol Myers Squibb, for the treatment of adults
The FDA recently approved a new infused therapy, Xenpozyme (olipudase-alfa) from Genzyme , for pediatric and adult patients with Acid Sphingomyelinase Deficiency (ASMD), a rare
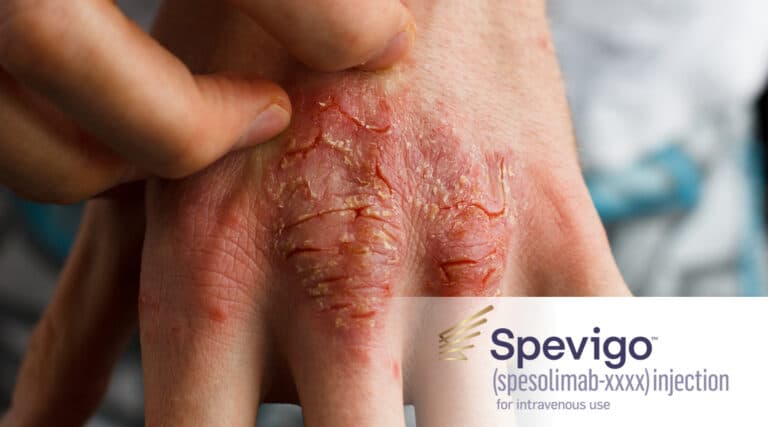
The FDA recently approved a novel infused therapy, Spevigo (spesolimab-sbzo) from Boehringer Ingelheim), with an indication for the treatment of generalized pustular psoriasis (GPP) flares

The FDA recently approved the fourth biosimilar to Genentech’s Avastin, Vegzelma (bevacizumab-adcd) from Celltrion USA. Vegzelma is an infused therapy administered by a healthcare professional.

The FDA recently approved a novel infused therapy, Terlivaz (terlipressin) from Mallinckrodt, with an indication for hepatorenal syndrome (HRS) with rapid reduction in kidney function. There are

The FDA recently approved a BLA for Rolvedon (eflapegrastim-xnst) from Spectrum Pharmaceuticals. It has an indication to decrease the incidence of infection, as manifested by febrile
We use cookies to improve your experience on our site. By using our site, you consent to cookies.
Manage your cookie preferences below:
Essential cookies enable basic functions and are necessary for the proper function of the website.
Statistics cookies collect information anonymously. This information helps us understand how visitors use our website.
Marketing cookies are used to follow visitors to websites. The intention is to show ads that are relevant and engaging to the individual user.
You can find more information about our Cookie Policy and Privacy Policy.