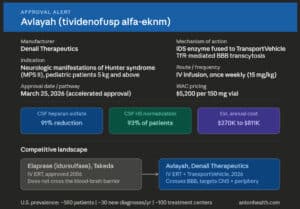
The FDA recently approved the fourth biosimilar to Genentech’s Avastin, Vegzelma (bevacizumab-adcd) from Celltrion USA. Vegzelma is an infused therapy administered by a healthcare professional.
Vegzelma is indicated for the treatment of patients with:
- metastatic colorectal cancer (mCRC);
- recurrent or metastatic non-squamous non-small cell lung cancer (nsNSCLC);
- recurrent glioblastoma (GBM);
- metastatic renal cell carcinoma (mRCC);
- persistent, recurrent, or metastatic cervical cancer (CC);
- epithelial ovarian, fallopian tube, or primary peritoneal cancer.
Vegzelma is Celltrion’s third oncology biosimilar approved for use in the U.S., following the approval of Truxima (rituximab-abbs) and Herzuma (trastuzumab-pkrb).
Approval did not include an interchangeable designation.
According to IQVIA, U.S. annual sales for bevacizumab for the 12 months ended February 2022 were $2.6 billion, $1.6 billion of which represented biosimilar sales.
Celltrion did not disclose pricing at this time.
————————————————————————————–
Celltrion USA Receives U.S. FDA Approval for its Oncology Biosimilar Vegzelma (bevacizumab-adcd) for the Treatment of Six Types of Cancer
# Vegzelma is Celltrion’s third oncology biosimilar to receive approval from the U.S. FDA
# Vegzelma offers U.S. patients living with multiple types of cancer a new, safe, and effective treatment option
September 28, 2022 — CHICAGO–(BUSINESS WIRE)–Celltrion USA announced that the U.S. Food and Drug Administration (FDA) has approved Vegzelma (bevacizumab-adcd), a biosimilar to Avastin (bevacizumab), for the treatment of six types of cancer: metastatic colorectal cancer; recurrent or metastatic non-squamous non-small cell lung cancer (nsNSCLC); recurrent glioblastoma; metastatic renal cell carcinoma; persistent, recurrent, or metastatic cervical cancer; and epithelial ovarian, fallopian tube, or primary peritoneal cancer.
“Biosimilars have been used in many disease areas including oncology and have shown to be safe and effective while lowering the drug cost and increasing the access to more patients around the world,” said Professor Claire Verschraegen, Director of the Division of Medical Oncology at the Ohio State University Comprehensive Cancer Center, Columbus, OH. “With the availability of biosimilars such as Vegzelma in the U.S., oncologists will have additional treatment options for patients across multiple cancer types.”
The FDA approval of Vegzelma was based on the totality of evidence, including the pivotal phase III trial in patients with metastatic or recurrent nsNSCLC. Results showed that as a first-line treatment, Vegzelma is highly similar to the reference product in terms of efficacy, safety and pharmacokinetics.
“The approval of Vegzelma is an important milestone in the U.S. which adds to our growing portfolio of oncology treatments and marks an important step forward in expanding access to cancer care,” said Jaeik Shim, Chief Operating Officer at Celltrion USA. “As a leading force in the global biopharmaceutical industry, we look forward to working with payers and providers to make our product available to patients. With our high-quality and affordable biosimilar medicines, we plan to strengthen our presence in the U.S. and contribute to a more sustainable healthcare system for the future.”
Vegzelma is an anti-cancer monoclonal antibody treatment biosimilar to Avastin® (bevacizumab). Vegzelma is a recombinant humanized monoclonal antibody which binds to vascular endothelial growth factor (VEGF), the key driver of vasculogenesis and angiogenesis, and thereby inhibits the binding of VEGF to its receptors Flt-1 (VEGFR-1), and kinase insert domain receptor (KDR) (VEGFR-2), on the surface of endothelial cells. In the U.S.,