……..catching up on FDA approvals
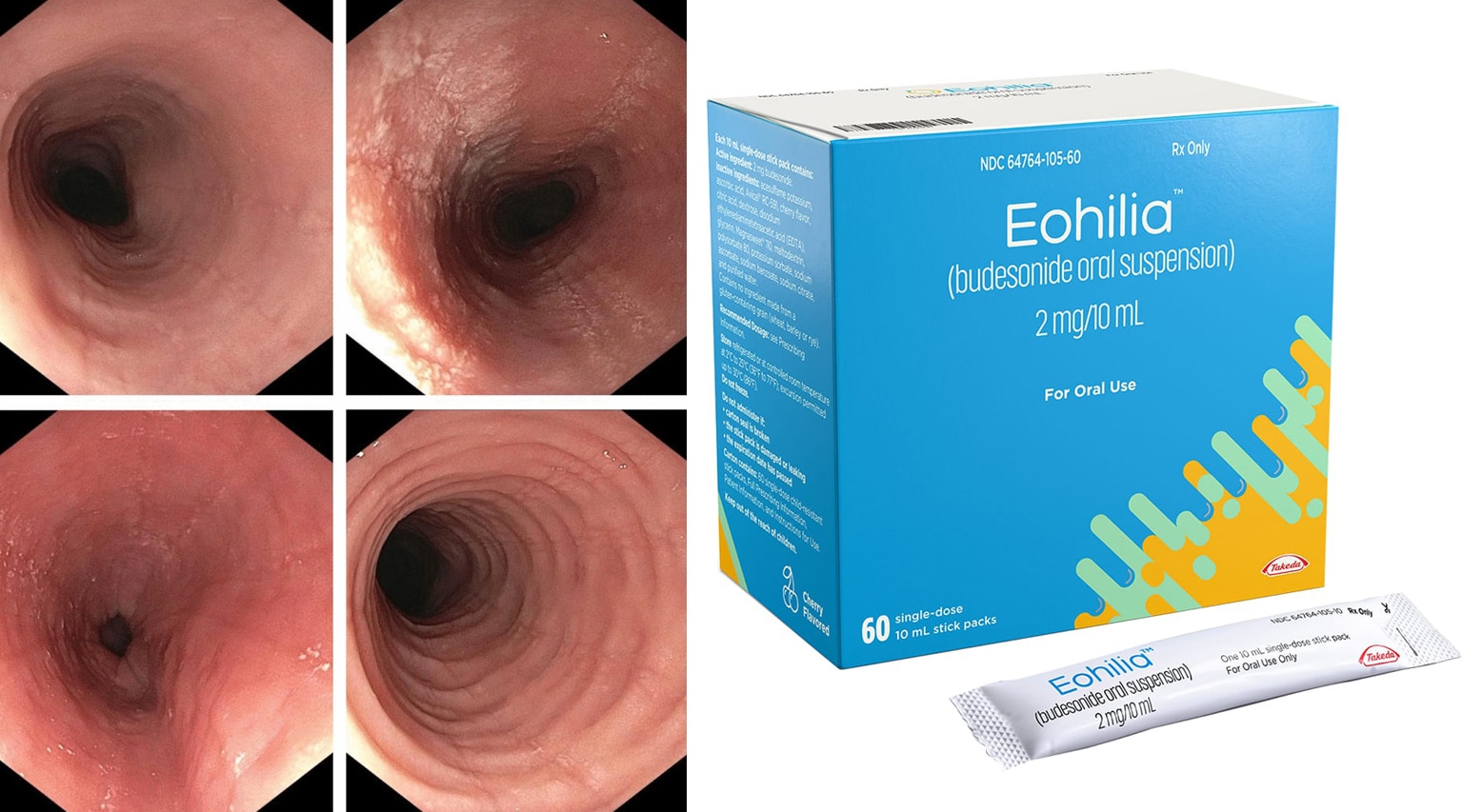
The FDA recently approved a new form of Eohilia (budesonide oral suspension) from Takeda , the first and only FDA-approved oral therapy for patients 11 years and older with eosinophilic esophagitis (EoE). Budesonide was first approved by the FDA under the brand name Pulmicort in 1997 as an oral inhalation formulation for the treatment of asthma.
EoE is a chronic disease that can significantly impact patients causing esophageal inflammation and intermittent symptoms of choking and difficult or painful swallowing. The largest study in the U.S. found the prevalence of EoE to be 57/100,000, or approximately 152,000 cases.
Eohilia is given over a 12-week course in single-dose 2 mg/10 ml stick packs, taken twice daily. It has a wholesale acquisition cost of $1,875 per month. Eohilia will compete with Dupixent which costs around $3,800 for two prefilled pens, which are taken either once a week or once every other week for EoE. Eohilia is supplied in a carton containing 60 single-dose stick packs.
Takeda did not disclose distribution details. As Eohilia is limited to 12 weeks of treatment and has a relatively low cost compared to most specialty pharmacy therapies, it would not be surprising to see this therapy available through open access.
Click here for prescribing information
————————————————————————————-
FDA Approves Takeda’s EOHILIA (budesonide oral suspension), the First and Only Oral Treatment in the U.S. for Eosinophilic Esophagitis (EoE)
— 12 Weeks of Treatment with EOHILIA May Address Significant Unmet Needs of Patients 11 Years of Age and Older
— EoE Is a Chronic Disease That Can Significantly Impact Patients, with Esophageal Inflammation and Intermittent Symptoms of Choking and Difficult or Painful Swallowing