FDA has now approved four gene therapies within the past ninety days.
That’s got to be a record!
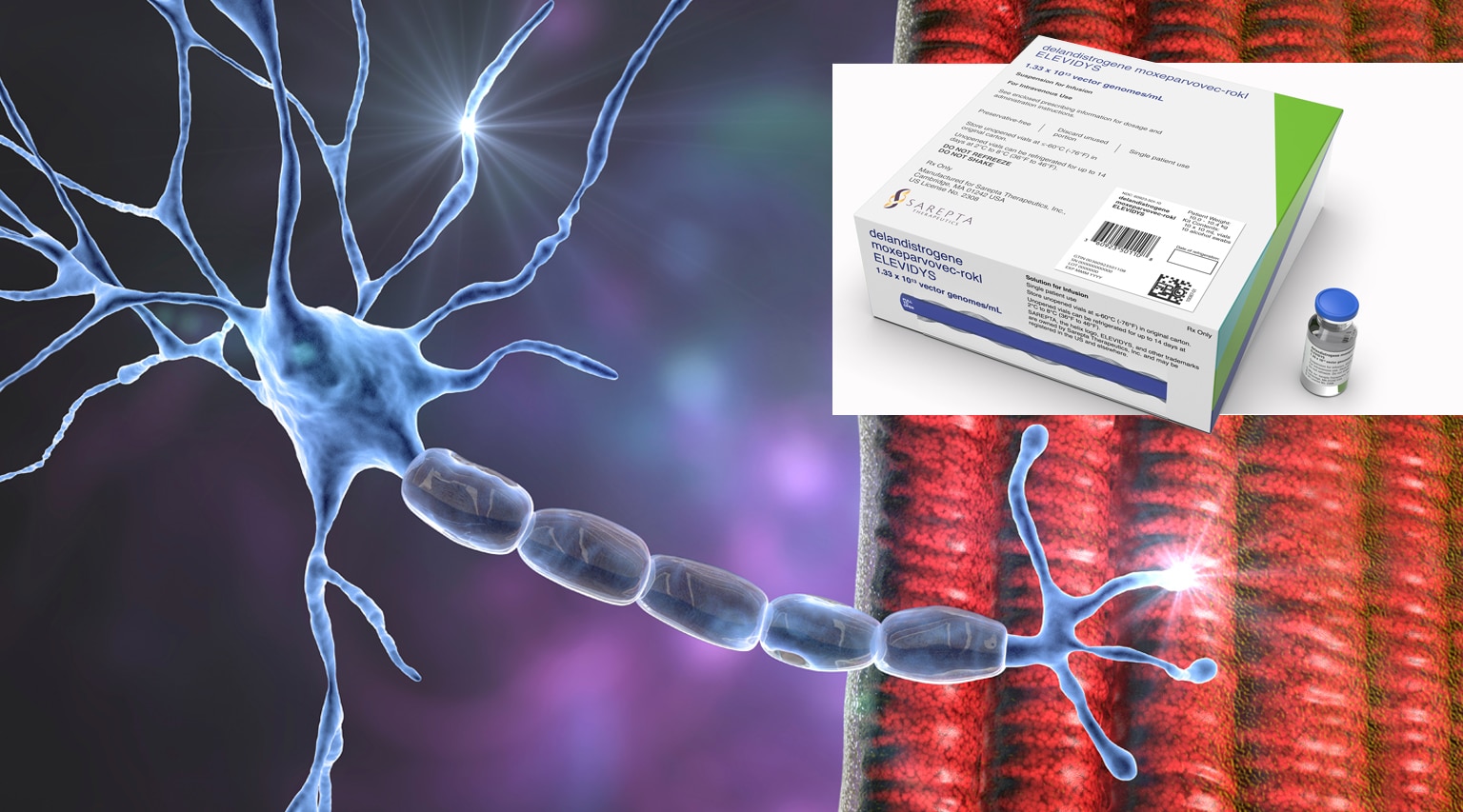
The FDA recently approved a new infused gene therapy, Elevidys (delandistrogene moxeparvovec-rokl) from Sarepta Therapeutics. Elevidys is administered as a single-dose intravenous infusion and is the first and only gene therapy approved for Duchenne. It is an adeno-associated virus-based gene therapy for the treatment of ambulatory pediatric patients aged 4 through 5 years with Duchenne muscular dystrophy (DMD) with a confirmed mutation in the DMD gene. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
Elevidys addresses the root genetic cause of Duchenne – mutations in the dystrophin gene that result in the lack of dystrophin protein – by delivering a gene that codes for a shortened form of dystrophin to muscle cells known as Elevidys micro-dystrophin. This accelerated approval is based on an increase in Elevidys micro-dystrophin protein expression in skeletal muscle.
Elevidys is the sixth approved therapy in Duchenne and the first therapy to replace the missing dystrophin protein by using a modified smaller version of the dystrophin gene, called micro-dystrophin, to produce a modified micro-dystrophin protein .Duchenne is a fatal genetic disorder that slowly robs patients of their muscle strength.
The therapy was approved without a black box warning but alerts were cited for Acute Serious Liver Injury, Immune-mediated Myositis, and Myocarditis.
Sarepta announced that Elevidys will launch at a one-time treatment cost of $3.2 million.
Details related to distribution and logistics were not released at time of approval, but industry sources have confirmed that at least one specialty pharmacy will be selected as the company’s specialty distribution partner.
CLICK HERE for full prescribing information
——————————————————————————————-
Sarepta Therapeutics Announces FDA Approval of Elevidys, the First Gene Therapy to Treat Duchenne Muscular Dystrophy
CAMBRIDGE, Mass.–(BUSINESS WIRE)–Jun. 22, 2023– Sarepta Therapeutics, Inc. (NASDAQ: SRPT), the leader in precision genetic medicine for rare diseases, today announced U.S. Food and Drug Administration (FDA) accelerated approval of Elevidys (delandistrogene moxeparvovec-rokl), …………