FDA Approves Last Specialty Tx of 2020 – Orgovyx
The FDA approved the last specialty therapy for 2020 just in time for Christmas. The new oral drug, Orgovyx from four-year-old Myovant Sciences, is indicated

The FDA approved the last specialty therapy for 2020 just in time for Christmas. The new oral drug, Orgovyx from four-year-old Myovant Sciences, is indicated
December 29, 2020Winter Garden, FL More catching up —The FDA approved another specialty therapy earlier this month. The new approval is Margenza (margetuximab-cmkb) from MacroGenics and

There really hasn’t been a lot of good news in the specialty pharmacy industry this year. Demand has been down due to the virus. Increasing

This year saw few specialty pharmacy acquisitions…. but that just changed with the announcement by Centene Corporation that it has acquired PantherRx Rare Pharmacy. Terms

Someone must have lit a fire under the FDA new drug approval elves in the last few weeks as the activity level there suddenly boomed.

We ran across a press release recently that looked a tad odd. It appeared that the FDA had approved a new therapy, Alkindi Sprinkle from
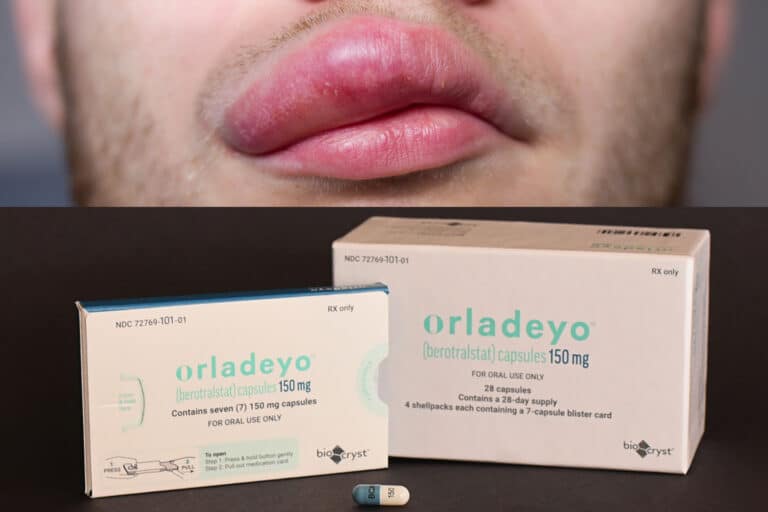
Earlier this month the FDA approved yet another orphan drug, Orladeyo (berotralstat) from BioCryst Pharmaceuticals. It obtained an indication for the prevention of hereditary angioedema

Ok, we know you are ready for more catch up….. and it ain’t the red stuff. The FDA recently approved yet another expensive specialty pharmacy

Ok, catch up time.Looking back a couple of weeks….. the FDA approved yet another rare/orphan therapy (which is mostly all they they’ve approved in 2020!).

With the emergence of CV-19 just about every health care provider has been scrambling to do something to preserve their patient relationships. A patient that